Angemeldet als:

Read more
Read less
Selected product solutions allow you to execute key methods and to process samples according to their unique requirements. Optimally matched equipment facilitates the acceleration and streamlining of processes and the reduction of costs, while securing reproducible, high-quality outcomes.
From producing target antigens up to cell line development, you benefit from optimal centrifugation, smart lab solutions, high-performance incubators, smart cold storage and reliable liquid handling tools. Eppendorf’s solutions provide excellence at every step!
Mehr erfahren
Weniger lesen
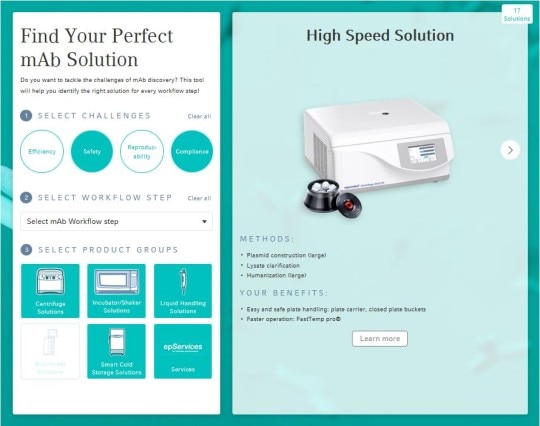
The most efficient way to tackle mAb discovery
This interactive tool will help you find the right solution for your mAb challenge.
By selecting your specific challenge and/or workflow step you can quickly and easily filter and browse available product groups and services.
Product Solutions
Compliance
Efficiency
Reproducibility
FACILITATE COMPLIANCE PROCESSES
Mehr erfahren
Weniger lesen
GAIN SPEED AND EFFICIENCY
Mehr erfahren
Weniger lesen
Improve reproducibility and sample safety
Mehr erfahren
Weniger lesen

All mAb product solutions

Centrifugation Solutions
Optimal separation of your samples to produce fast and reliable results.
Incubator & Shaker Solutions
Let your cells grow under reliable and reproducible cultivation conditions.

Liquid Handling Solutions
Transfer liquids, bacteria and cells with reliable pipettes at highest accuracy and precision.

Bioprocess Solutions
Set up reproducible and scalable processes from bench to pilot and production.

Smart Cold Storage Solutions
Identify, track and store your samples. Monitor conditions, processes and freezer performance.

Digital Lab Solutions
Increase your productivity when handling routine tasks with smart lab management software solutions.
Request your individual support
Request your individual support
Your own dedicated expert assists you in finding the perfect match for your specific mAb workflow. Get your individual consultation!
Mehr erfahren
Weniger lesen











