MENU
AE | USD
AE | USD
-
- Toutes les centrifugeuses
- Centrifugeuses de paillasse
- Centrifugeuses au sol
- Centrifugeuses réfrigérées
- Microcentrifugeuses
- Centrifugeuses polyvalentes
- Centrifugeuses haute vitesse
- Ultracentrifugeuses
- Concentrateur
- Consommables pour centrifugeuses haute vitesse et ultracentrifugeuses
- Tubes à centrifuger
- Plaques de centrifugeuse
- Logiciel de gestion d’appareils
- Gestion des échantillons et des informations
-
- Pipettes, distributeurs et dispositifs de manipulation de liquides automatisée
- Pipettes mécaniques
- Pipettes électroniques
- Pipettes multicanaux
- Pipettes et distributeurs à déplacement positif
- Pointes de pipette
- Distributeurs sur flacon
- Auxiliaires de pipetage
- Accessoires pour pipettes et distributeurs
- Pipetage automatisé
- Consommables d’automatisation
- Accessoires d’automatisation
- Services pour pipettes et distributeurs
Sorry, we couldn't find anything on our website containing your search term.
Vous vous apprêtez à quitter ce site.
Veuillez noter que votre panier actuel n’est pas encore enregistré et ne pourra pas être affiché sur le nouveau site ou lors de votre prochaine visite. Si vous souhaitez enregistrer votre panier, veuillez vous connecter sur votre compte.
Sorry, we couldn't find anything on our website containing your search term.
- Accueil
- Produits
- Bioprocessus
- Bioprocess Equipment for Commercial Manufacturing
- DASware® control plus
DASware® control plus
DASware control plus single

DASware control plus
• DASware control software with 21 CFR Part 11 and EudraLex Volume 4 Annex 11 compatibility, providing User Authentication and Access Control, electronic records, and electronic signatures
• DASware control software with 21 CFR Part 11 and EudraLex Volume 4 Annex 11 compatibility, providing User Authentication and Access Control, electronic records, and electronic signatures
Informations produit
DASware control plus est un logiciel SCADA pour les bioprocédés, conforme avec le Code des règlements fédéraux, Titre 21, Partie 11 et EudraLex Volume 4 Annexe 11 pour une utilisation en environnement de production clinique et commerciale Il peut être utilisé pour le contrôle des bioprocédés à l’aide des systèmes de contrôle des bioréacteurs Eppendorf, tels que le dispositif de contrôle BioFlo 320 à l’échelle du laboratoire. DASware control plus permet la surveillance et le contrôle individuels des bioprocédés parallèles, l’automatisation des processus et l’analyse avancée des données, offrant une performance maximale de la conception expérimentale jusqu’à l’obtention des résultat. Il offre des fonctionnalités adaptées à l’optimisation de la gestion des documents électroniques, à la sécurisation des signatures électroniques et à l’intégrité de la chronologie des événements.Vous trouverez plus d’informations à télécharger au bas de cette page
Produits (1)
1 Produits
|
|
Afficher plus de produits
Ajouté à votre panier
Informations produit
Caractéristiques
Informations produit
DASware control plus: il a été spécialement conçu pour être compatible avec le Code des règlements fédéraux, Titre 21, Partie 11 et EudraLex Volume 4 Annexe 11, afin d’aider les utilisateurs à se conformer aux critères définis par ces normes.
• Authentification de l’utilisateur et contrôle d’accès : conforme aux exigences des BPF pour empêcher tout accès non autorisé et maintenir l’intégrité des données.
• Enregistrements électroniques : assurer le suivi et la traçabilité des actions critiques au sein d’une chronologie des événements, garantissant une visibilité complète sur l’intégrité des données et la conformité réglementaire.
• Signatures électroniques avec confidence : assurer l’authenticité et la fiabilité de la documentation essentielle dans vos opérations de bioprocédés.
• Gestion efficace des données : extraction et stockage des données humaines et électroniques, lisibles par le système, tout en respectant des normes strictes d’intégrité des données.
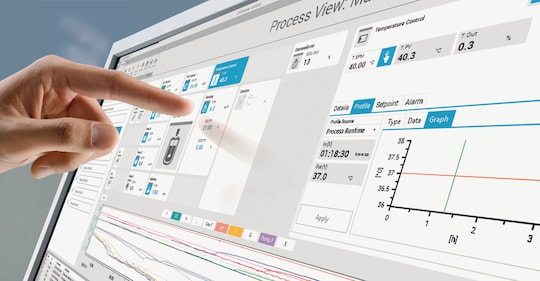
Systèmes de bioréacteurs et solutions logicielles : intégrés pour assurer le contrôle, l’automatisation et l’analyse des bioprocédés
DASware control est la solution logicielle centrale d’Eppendorf pour les bioprocédés. Au-delà de la surveillance et du contrôle des bioprocédés, il permet l’intégration optimale des systèmes de bioprocédés, des logiciels de contrôle, des outils d’analyse des données et des solutions d’automatisation pour constituer une solution complète de bioprocédés en amont.
• Surveillance et contrôle des processus en parallèle
• Complexité réduite grâce à l’utilisation d’une solution logicielle centrale
• Intégration facile de dispositifs tels que des capteurs tiers
• Partage simplifié et analyse améliorée des données grâce à une connexion fiable au logiciel BioNsight® cloud
Eppendorf Bioprocess Products – application in GMP regulated environment
Download document to find out which features you can expect at minimum from Eppendorf products that are deemed suitable or compatible for GMP.
• Authentification de l’utilisateur et contrôle d’accès : conforme aux exigences des BPF pour empêcher tout accès non autorisé et maintenir l’intégrité des données.
• Enregistrements électroniques : assurer le suivi et la traçabilité des actions critiques au sein d’une chronologie des événements, garantissant une visibilité complète sur l’intégrité des données et la conformité réglementaire.
• Signatures électroniques avec confidence : assurer l’authenticité et la fiabilité de la documentation essentielle dans vos opérations de bioprocédés.
• Gestion efficace des données : extraction et stockage des données humaines et électroniques, lisibles par le système, tout en respectant des normes strictes d’intégrité des données.
Systèmes de bioréacteurs et solutions logicielles : intégrés pour assurer le contrôle, l’automatisation et l’analyse des bioprocédés
DASware control est la solution logicielle centrale d’Eppendorf pour les bioprocédés. Au-delà de la surveillance et du contrôle des bioprocédés, il permet l’intégration optimale des systèmes de bioprocédés, des logiciels de contrôle, des outils d’analyse des données et des solutions d’automatisation pour constituer une solution complète de bioprocédés en amont.
• Surveillance et contrôle des processus en parallèle
• Complexité réduite grâce à l’utilisation d’une solution logicielle centrale
• Intégration facile de dispositifs tels que des capteurs tiers
• Partage simplifié et analyse améliorée des données grâce à une connexion fiable au logiciel BioNsight® cloud
Eppendorf Bioprocess Products – application in GMP regulated environment
Download document to find out which features you can expect at minimum from Eppendorf products that are deemed suitable or compatible for GMP.


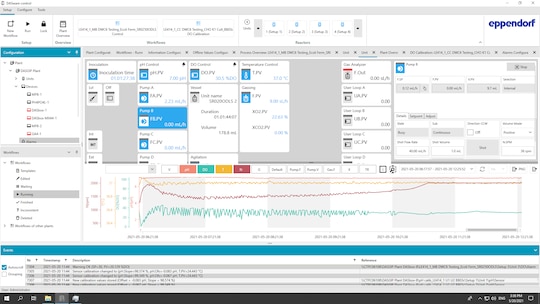
Bénéficier d’un contrôle puissant des bioprocédés
• Efficacité : étalonnage parallèle des capteurs, gestion intelligente des recettes et automatisation améliorée des scripts.
• Performance : jusqu’à 8/16/24 récipients en parallèle, un outil performant de visualisation graphique et une gestion avancée des données.
• Flexibilité : vues configurables et fonctions définies par l’utilisateur ; mises à jour matérielles et logicielles sur le terrain.
• Fiabilité : gestion des utilisateurs et intégrité des données, journal des événements et fonctions d’alarme.
• Normes réglementaires : Compatible avec le Code des règlements fédéraux, Titre 21, Partie 11 et EudraLex Volume 4 Annexe 11. Si vous avez besoin d’un logiciel de contrôle des bioprocédés à des fins de R&D, découvrez DASware control.


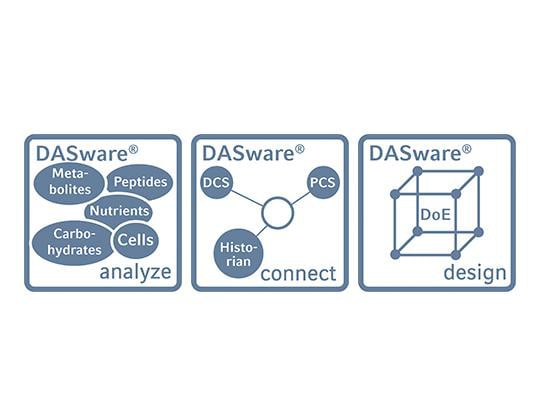

Développez vos capacités avec la suite logicielle DASware
DASware design, analyze et connect sont des compléments en option au logiciel DASware control SCADA.
• Mise en œuvre du concept de plan d’expériences (DoE) via un générateur de plans d’expériences factoriels complets ou via l’importation de plans d’expériences issus d’outils tiers
• Intégration optimale de dispositifs de laboratoire externes au bioréacteur pour l’automatisation des processus et les boucles de contrôle de rétroréaction
• Intégration dans les systèmes de contrôle de processus et les historiques d’entreprise hérités pour un accès à l’ensemble de l’entreprise à toutes les données pertinentes relatives aux bioprocédés




Vos données, à portée de main
Les données relatives aux bioprocédés peuvent être transférées de manière optimale du logiciel DASware control plusau logiciel BioNsight cloud.
• Permet la surveillance et l’analyse à distance.
• Permet la contextualisation des données entre les appareils, les cycles et les sites.
• Préparation des données pour une analyse reposant sur l’I


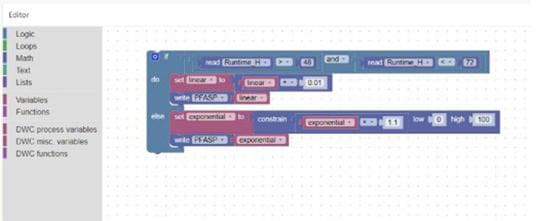
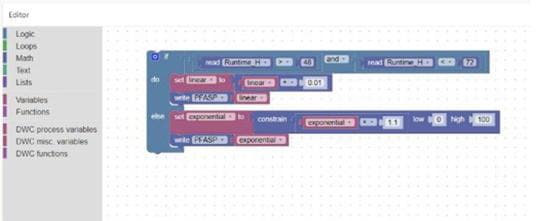
Personnalisez facilement votre contrôle de processus grâce à la programmation graphique
• Facilité d’utilisation : l’interface « glisser-déposer » permet aux utilisateurs de créer et de modifier des flux de travail en reliant visuellement des blocs fonctionnels, ce qui réduit la complexité par rapport à un script ou à un codage traditionnels.
• Exactitude améliorée : les flux de travail visuels aident les utilisateurs à rapidement identifier les erreurs de logique ou de séquence, limitant ainsi les interruptions de processus et améliorant l’exactitude globale.
• Accessibilité améliorée : les utilisateurs peuvent rapidement maîtriser les flux de travaux et les mettre en œuvre sans délai, ce qui augmente l’efficacité opérationnelle et la capacité d’adaptation aux modifications de processus.
• Transparence : l’enregistrement automatique de toutes les modifications assure la conformité, tandis que l’historique des révisions facilite les audits.


Lire plus
Lire moins
Caractéristiques
- Contrôle de processus en parallèle avec contrôle individuel de chaque récipient
- Fonction de lot intégrée pour la gestion des processus et des recettes
- Idéal pour la conception d’expériences (DoE)
- Procédures parallèles de calibrage et de nettoyage
- Vues personnalisées et fonctions définies par l’utilisateur
- Exportation automatique de données, génération de rapports et de graphiques pour Microsoft® Excel®
- Banque de données professionnelle avec accès géré (Postgre SQL Server)
- Communication OPC pour une intégration facile avec des équipements tiers utilisant DASware connect et DASware analyze
- Analyse intégrée des valeurs hors ligne, des valeurs calculées en ligne et de la notification des alarmes
- Connectivité et transfert de données fiables vers BioNsight cloud
- Pack IQ/OQ disponible