MENU
BR | BRL
BR | BRL
-
- All Centrifuges
- Benchtop Centrifuges
- Floor-Standing Centrifuges
- Refrigerated Centrifuges
- Microcentrifuges
- Multipurpose Centrifuges
- High-Speed Centrifuges
- Ultracentrifuges
- Concentrator
- High-Speed and Ultracentrifuge Consumables
- Centrifuge Tubes
- Centrifuge Plates
- Device Management Software
- Sample and Information Management
-
- All Pipettes, Dispensers & Automated Liquid Handlers
- Mechanical Pipettes
- Electronic Pipettes
- Multi-Channel Pipettes
- Positive Displacement Pipettes & Dispensers
- Pipette Tips
- Bottle-Top Dispensers
- Pipette Controllers
- Dispenser & Pipette Accessories
- Automated Pipetting
- Automation Consumables
- Automation Accessories
- Liquid Handler & Pipette Services
Sorry, we couldn't find anything on our website containing your search term.
Sorry, we couldn't find anything on our website containing your search term.

Webinar
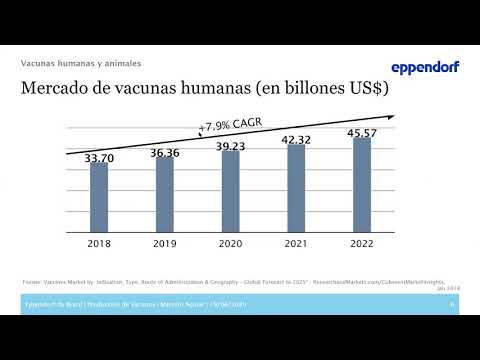
Production of vaccines – bioprocess development and scale-up
In these exceptional and unprecedented times, many companies are working eagerly on the development of a vaccine against COVID-19. New bioprocess technologies, such as single-use equipment and process automation, open up new possibilities for quality control and validation.
This is particularly important in GMP-regulated environments, such as in the development and manufacture of influenza vaccines. When facing a pandemic outbreak, developing new processes and rapidly increasing the volume of clinical production are essential for efficient development of new vaccines.
Date and time: These webinars took place on June 9 and 19, 2020.
Speaker: Marcelo Aguiar Msc., Zone Sales Director, Eppendorf do Brasil Ltda.
Languages: The recorded webinars are only available in Spanish.
Watch here the recordings of the webinars:
Date and time: These webinars took place on June 9 and 19, 2020.
Speaker: Marcelo Aguiar Msc., Zone Sales Director, Eppendorf do Brasil Ltda.
Languages: The recorded webinars are only available in Spanish.
Watch here the recordings of the webinars:
Read more
Videos not loading, because cookies have been rejected. Change your

Videos not loading, because cookies have been rejected. Change your