MENU
DE | EUR
DE | EUR
No results found
Search Suggestions
What Should Be Considered when Pipetting PCR Mixtures?
Lab Academy
- Molecular Biology
- Pipettes
- Pipette Tips
- PCR Cyclers
- Amplification & PCR
- Essay
For successful amplification reactions, it is necessary that the individual reaction components are present in the correct concentration in each preparation. In addition, it is important that no contamination occurs.
Especially when many reactions have to be set-up, it has been established to prepare a so-called master mix instead of pipetting each reagent separately into each vessel. Pre-configured mixes are commercially available, in which only sample-specific components (primer) and water are added. Alternatively, the master mix can be prepared by yourself. In both variants, the mixture is distributed to each PCR vessel without a template and the individual DNA sample is added separately at the end.
Using a master mix has several advantages: First, the number of single pipetting steps is reduced. In this way, both the risk of user errors during pipetting and the risk of contamination are minimized and, of course, time is saved. In principle, the pipetting accuracy is also higher, since larger volumes are dosed. This is easy to understand when checking the technical data of pipettes: The smaller the dosed volume, the higher the deviations can be (table 1). The fact that all preparations come from the same vessel has a positive effect on homogeneity (if well mixed). This also improves the reproducibility of the experiments.
Using a master mix has several advantages: First, the number of single pipetting steps is reduced. In this way, both the risk of user errors during pipetting and the risk of contamination are minimized and, of course, time is saved. In principle, the pipetting accuracy is also higher, since larger volumes are dosed. This is easy to understand when checking the technical data of pipettes: The smaller the dosed volume, the higher the deviations can be (table 1). The fact that all preparations come from the same vessel has a positive effect on homogeneity (if well mixed). This also improves the reproducibility of the experiments.
Read more
Read less

When preparing the master mix, at least 10 % extra volume should be added (e.g. if 10 preparations are required, calculate on the basis of 11), so that even the last vessel filled properly. In this way, (slight) pipetting inaccuracies, and the effect of sample loss when dosing detergent-containing solutions can be compensated. Detergents are contained in enzyme solutions such as polymerases and master mixes, causing foam formation and residues on the inner surface of normal pipette tips.
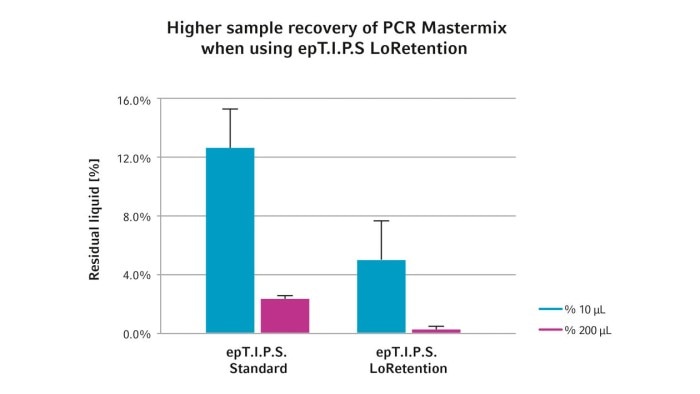
Depending on the application and the type of liquid to be dispensed, the correct pipetting technique (1) should be chosen and the appropriate equipment selected (2). For solutions containing detergents, a direct displacement system or the so-called "low retention" pipette tips as an alternative for air-cushion pipettes are recommended. The effect of epT.I.P.S.® LoRetention is based on a particularly hydrophobic surface. Liquids containing detergents do not leave a residue film on the inside and outside, so that the loss of solution can be minimized (Figure 1).
Depending on the application and the type of liquid to be dispensed, the correct pipetting technique (1) should be chosen and the appropriate equipment selected (2). For solutions containing detergents, a direct displacement system or the so-called "low retention" pipette tips as an alternative for air-cushion pipettes are recommended. The effect of epT.I.P.S.® LoRetention is based on a particularly hydrophobic surface. Liquids containing detergents do not leave a residue film on the inside and outside, so that the loss of solution can be minimized (Figure 1).
Read more
Read less
Besides the exact dosing of all components, it is also important that no contamination of the preparations occurs. It is not sufficient to use consumables of high purity, because the pipetting process in an air cushion pipette can produce aerosols that remain in the pipette. The DNA that may be contained in the aerosol can be transferred from one sample to the next in the following pipetting step and thus lead to contamination. The direct displacement systems mentioned above may also minimize this risk. For air-cushion pipettes it makes sense to use filter tips to protect the pipette cone by retaining splashes, aerosols, and biomolecules.
References:
[1] Userguide Pipette families 112928_Userguide
[2] Pipettes and dispensers 112927_Userguide
References:
[1] Userguide Pipette families 112928_Userguide
[2] Pipettes and dispensers 112927_Userguide
Read more
Read less

