MENU
US | USD
US | USD
-
- All Centrifuges
- Benchtop Centrifuges
- Floor-Standing Centrifuges
- Refrigerated Centrifuges
- Microcentrifuges
- Multipurpose Centrifuges
- High-Speed Centrifuges
- Ultracentrifuges
- Concentrator
- High-Speed and Ultracentrifuge Consumables
- Centrifuge Tubes
- Centrifuge Plates
- Device Management Software
- Sample and Information Management
-
- All Pipettes, Dispensers & Automated Liquid Handlers
- Mechanical Pipettes
- Electronic Pipettes
- Multi-Channel Pipettes
- Positive Displacement Pipettes & Dispensers
- Pipette Tips
- Bottle-Top Dispensers
- Pipette Controllers
- Dispenser & Pipette Accessories
- Automated Pipetting
- Automation Consumables
- Automation Accessories
- Liquid Handler & Pipette Services
Sorry, we couldn't find anything on our website containing your search term.
Sorry, we couldn't find anything on our website containing your search term.
- Home
- Products
- Temperature Control and Mixing
- Accessories
- ThermoTop®
ThermoTop®
Reduce condensation by using a heated lid

ThermoTop provides heating from top to reduce condensation within lid of tubes
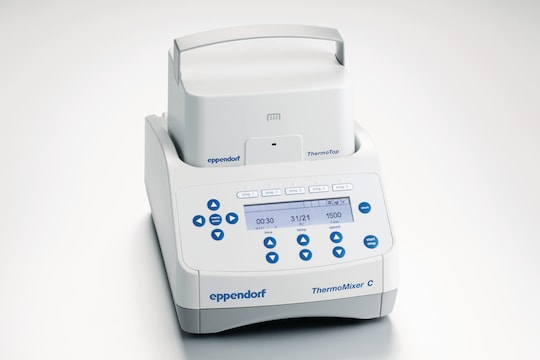
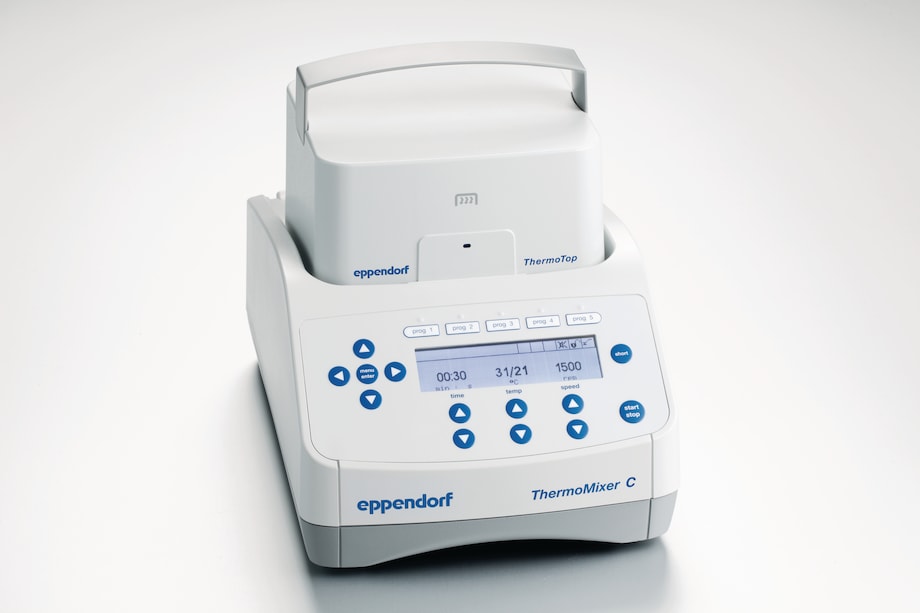
Eppendorf ThermoMixer C with ThemoTop

When the ThermoTop is placed on the device, it begins heating immediately. The temperature optimally adapts to the test temperature to ensure that it does not negatively affect the sample. Condensation droplets have no opportunity to form.
ThermoStat C equipped with ThermoTop

Eppendorf ThermoStat C with ThermoTop: Reduce condensation within the sample vials
ThermoMixer C SmartBlock_ThermoTop

Eppendorf ThermoMixer C with ThermoTop
ThermoMixer C total_ThermoTop
Counter heating needed? Safe samplehandling due to extra heat control by ThermoTop
Eppendorf ThermoStat C with ThermoTop

Condensation issues? By using the ThermoTop, you can minimize condensation in your tubes or plates for more reproducible results.
Reliable experiments by condens.protect®
Videos not loading, because cookies have been rejected. Change your

Product Information
Having non-regular enzymatic results after temperature incubation?The Eppendorf ThermoTop with condens.protect technology heated lid avoids condensation on tube lid and tube wall. It is compatible with ThermoMixer C/ ThermoStat C with SmartBlock plates and lab vessels up to 2 mL and ThermoMixer F 0.5/F1.5/F2.0/FP.
ThermoTop
You will find additional download material at the bottom of this page
Products (1)
Accessories (1)
1 Products
Price
$ 1,010.66
|
Show more Products
Added to Your Cart
Product Information
Applications
Features
Product Information
Tired of condensation in your tubes?
We combined the Eppendorf ThermoMixer® with the PCR cycler approach of a heated lid and generated the Eppendorf ThermoTop. The outstanding Eppendorf condens.protect® technology reliably prevents condensation within the tube lid – sample concentrations are consistent for the incubation time. In addition, the temperature homogeneity is further improved. Get optimal sample reaction conditions to produce optimal results.
Perfect condensation prevention when dealing with small volumes: The Eppendorf ThermoTop with condens.protect technology heated lid avoids condensation on tube lid and tube wall. It is compatible with ThermoMixer C/ ThermoStat C with SmartBlock plates and lab vessels up to 2 mL and ThermoMixer F 0.5/F1.5/F2.0/FP.
We combined the Eppendorf ThermoMixer® with the PCR cycler approach of a heated lid and generated the Eppendorf ThermoTop. The outstanding Eppendorf condens.protect® technology reliably prevents condensation within the tube lid – sample concentrations are consistent for the incubation time. In addition, the temperature homogeneity is further improved. Get optimal sample reaction conditions to produce optimal results.
Perfect condensation prevention when dealing with small volumes: The Eppendorf ThermoTop with condens.protect technology heated lid avoids condensation on tube lid and tube wall. It is compatible with ThermoMixer C/ ThermoStat C with SmartBlock plates and lab vessels up to 2 mL and ThermoMixer F 0.5/F1.5/F2.0/FP.
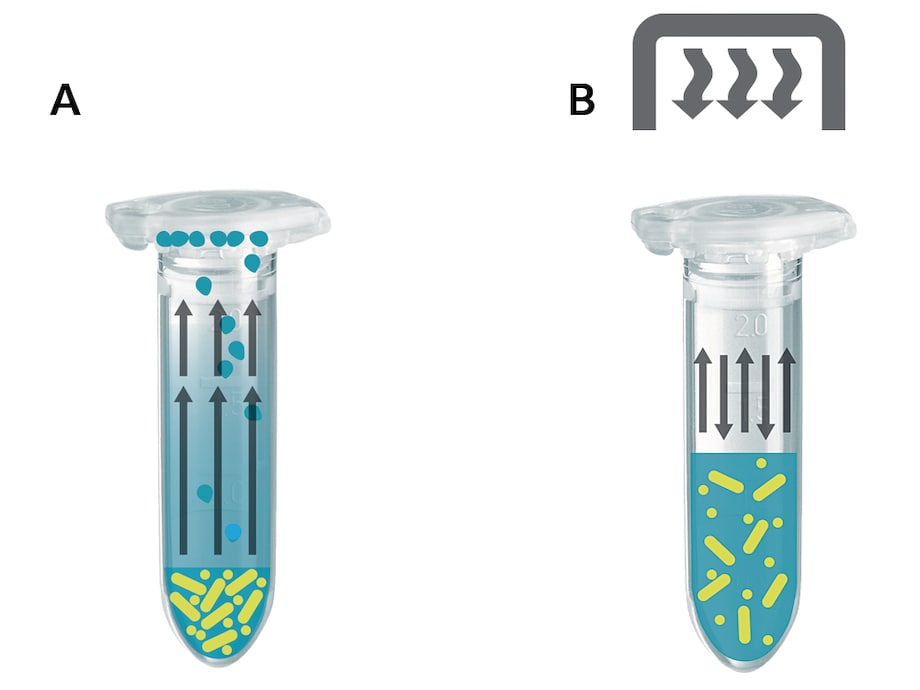
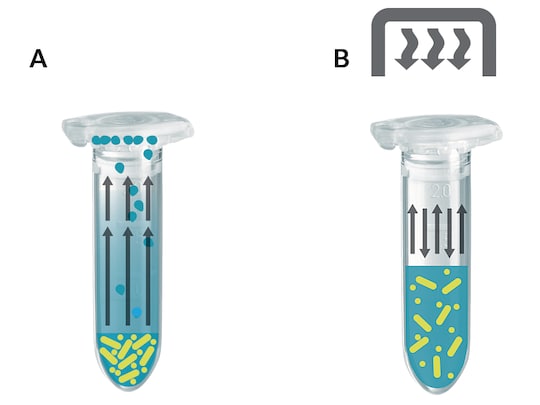
What happens in the tube?
Without the use of a heated lid, solvent molecules will evaporate to the gaseous phase during the heating process of a liquid sample and subsequently condensate against the cooler surfaces of lid and container wall. Hence, the volume of the sample is reduced while its concentration increases (fig. A).
Sensitive reactions may be influenced by this change in reaction conditions. In the case of very small sample volumes, the condensation process can leave such a small residual sample volume inside the reaction tube that the sample is no longer mixable. Under such conditions the biochemical reaction will be neither optimal nor reproducible.
When using the Eppendorf ThermoTop, the surfaces of the tube lids and upper tube walls are warmed in such a way as to prevent the formation of condensation effectively.
Thus, sample concentration remains stable, reaction conditions are strictly adhered to, and reproducibility of experiments is improved (fig. B).


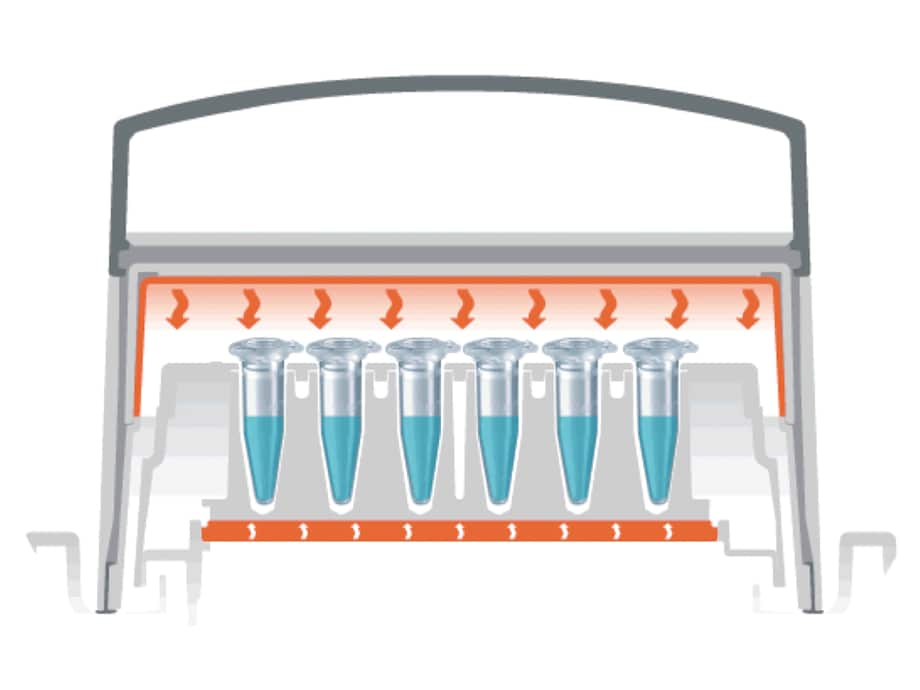
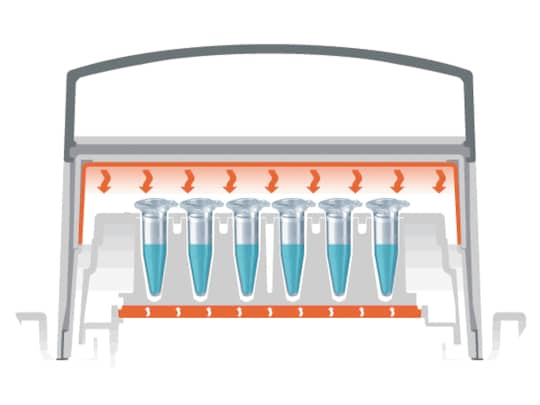
How does it work?
The ThermoTop impresses with a simple, wireless handling as well as the automatic recognition and operation. It is detected by the device as soon as it is positioned, via inductive signal transmission, and thermoregulation starts automatically. The temperature optimally adapts to the test temperature to ensure that it does not negatively affect the sample. Condensation droplets have no opportunity to form.




Where can I use it?
The ThermoTop is compatible with the ThermoMixer C, ThermoStat C, ThermoMixer F0.5/ F1.5/ F2.0 and ThermoMixer FP. Combined with the ThermoMixer C and the ThermoStat C, the ThermoTop can be used with thermoblocks for plates and lab vessels with a volume up to 2 mL. Compatible thermoblocks feature the condens.protect symbol.


Read More
Read Less
Applications
- All temperature control procedures for which the formation of condensation on the tube lid and wall should be reliably avoided
Features
- Efficient and reliable of condensation prevention for safe sample heating
- Get stable reaction conditions for optimal enzymatic performance within your samples
- Wireless connection with automatic recognition and operation for user-friendly handling
- Suitable for use with Eppendorf ThermoMixer® F0.5/F1.5/F2.0/FP as well as Eppendorf ThermoMixer C and Eppendorf ThermoStat C with Eppendorf SmartBlocks 0.5 mL, 1.5 mL, 2.0 mL, plates, PCR 96, and PCR 384 (not in combination with Eppendorf Transfer Rack or SmartExtender)
Downloads: ThermoTop®
-
English
You might also consider