Regarding suitability of Eppendorf bioprocess equipment in GMP-regulated applications, please reach out to your Eppendorf sales representative.
Stem Cell Bioprocessing

Read more
Read less
Webinar: Breaking boundaries in bioprocess development
Join experts from Hannover Medical School, Pluri Inc., and Eppendorf and find out more about unleashing the power of stirred-tank bioreactors for cell and gene therapies!

New to stem cell bioprocessing? Find your answers below.
What are bioreactors?
The success of stem cell bioprocessing relies on robust and reproducible culture conditions and processes. Bioreactors are vessels used to culture a broad range of cell types in precisely controlled conditions to provide optimal productivity, efficiency, and product quality. Bioreactors come in a range of sizes from milliliters to hundreds of liters. They can either be single-use or made of glass or stainless steel.
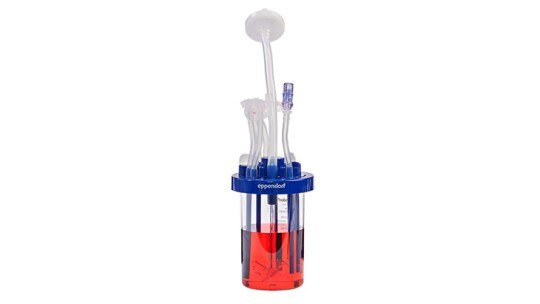
Read more
Read less
What are the advantages of bioreactors over cell culture flasks?
- Better reproducibility: In bioreactors you can monitor critical culture parameters including pH , Dissolved Oxygen (DO) , temperature and nutrient feeding and automatically adjust them by bioprocess monitoring and control software. Like this, culture conditions and as a result culture outcome can be better reproduced.
- Simpler scale-up: Many stem cell applications in cell therapy development, drug discovery, and lab-grown meat require high cell numbers. When using a bioreactor one can increase the size of the culture vessel to many liters and even more, unlike cell culture flasks where the number of vessels must be increased after a certain point. Using one big vessel instead of many small ones saves space and manual labor, thus simplifying the process of increasing the number of cultured cells.
Read more
Read less
Read more
Read less
How can I cultivate adherent stem cells in bioreactors?
The majority of stem cells are adherent cells and require a support matrix to grow. In 2D cultures, this means growing stem cells in dishes or flasks, however, these systems are often challenging to scale-up. Fortunately, there are many available options to grow adherent cells in bioreactors including:
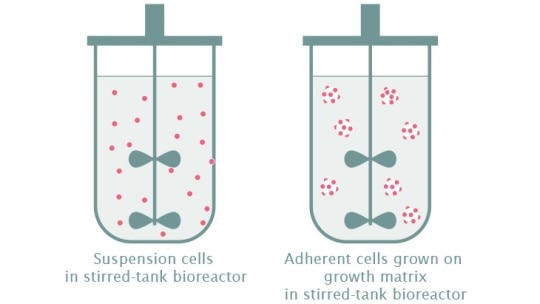
- Cell-only aggregates: This technique involves allowing cells to adhere to one another and form an aggregate. This technique is often used for the cultivation of induced pluripotent stem cells (iPSCs) in bioreactors.
- Microcarriers: Microcarriers are spherical beads, which are added to the culture medium and serve as a surface for adherent cells such as mesenchymal stem cells (MSCs) to attach and grow. Microcarriers are made of a variety of materials. They can have specific coatings to enhance cell attachment, yield and viability.
- Fibra-Cel® Disks: Disks made of polyester and polypropylene provide a growth support for adherent cells in both packed-bed bioreactors and in suspension. Due to their three-dimensionality and fibrous material, Fibra-Cel Disks allow for easy cell attachment, a high surface-to-volume-ratio, and provide a low-shear environment. This often results in increased total biomass compared to many microcarriers.
Read more
Read less
Read more
Read less
Can I also differentiate my stem cells in a bioreactor?
Read more
Read less
Can I cultivate iPSCs as well as MSCs in bioreactors?
Read more
Read less
How can I switch from stem cell culture in flasks and dishes to bioreactors?
Read more
Read less
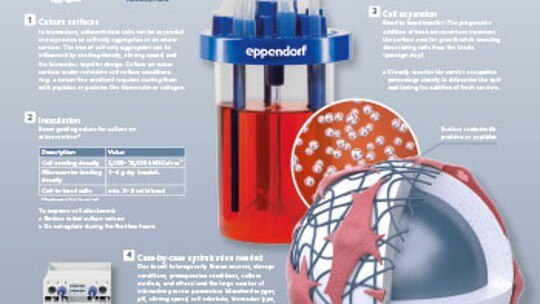
Infographic: Stem cell culture in bioreactors
Download our infographic for some tips to help you transfer your stem cell culture from dishes and flasks to bioreactors.
Interview: Increasing hiPSC yields
Robert Zweigerdt fromHannover Medical Schoolexplains how his groupconverted stem cell culture to bioreactors and which process optimization measures were crucial to increase the cell yield.

Panel discussion: Preparing your stem cell cultivation for commercialization
At what point in yourdevelopment should youconsider which cultivation method? How to switch from static to stirred-tank cultivation? Listen to experts from the Cell and Gene Therapy Catapult and Eppendorf to discover answers to these and more questions.

Read more
Read less
Stem cell expertise at Eppendorf
Read more
Read less
Choose the right bioreactor system
Bioreactor control stations
Read more
Read less
Single-use bioreactors
Read more
Read less
Read more
Read less
Stem cell bioprocessing for the experts: Optimize your stem cell culture in bioreactors
How can I increase the yield of my stem cell culture in bioreactors?
Read more
Read less
Read more
Read less
How can I control the critical process parameters in my bioreactor?
Bioprocess monitoring and control
Read more
Read less
Learn from biologics bioprocessing
Read more
Read less
Where can I find information about the cultivation of MSCs in bioreactors?
Read more
Read less
Read our application notes for some guidelines on how to culture MSCs in bioreactors



Where can I find information about the cultivation of iPSCs in bioreactors?
Read more
Read less
Read our application notes for some guidelines on how to culture hiPSCs in bioreactors


How can I produce stem cell-derived exosomes in bioreactors?
Read more
Read less
Join the stem cell community!
Read more
Read less













