MENU
MY | MYR
MY | MYR
No results found
Search Suggestions
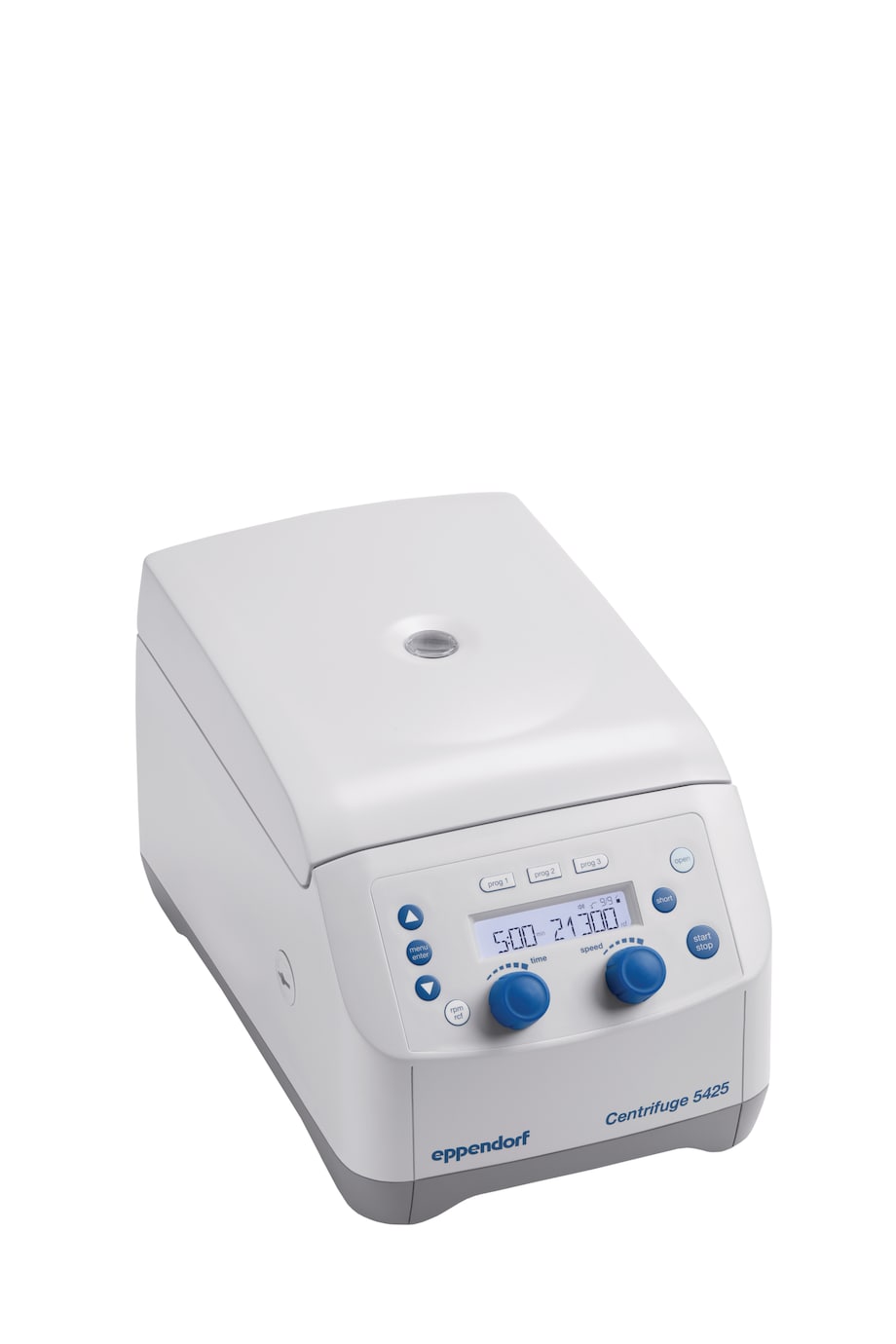
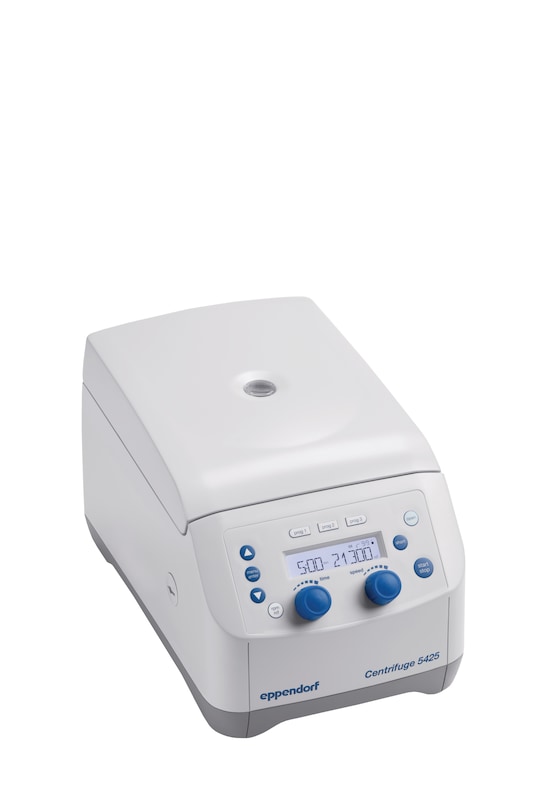
Centrifuge 5425

Medical Device registration in Malaysia: This Medical Device is registered under Act 737. This webpage content is intended for Healthcare Professionals only, not for general public. Click here for Malaysia Medical Device Authority (MDA) product registration listing.
All medical devices sold in Malaysia must carry local labelling as per requirement from the Medical Device Authority. To allow this to happen, the packaging of the non-refrigerated devices will be opened and resealed for labelling purposes.
Medical Device registration in Malaysia: This Medical Device is registered under Act 737. This webpage content is intended for Healthcare Professionals only, not for general public. Click here for Malaysia Medical Device Authority (MDA) product registration listing.
All medical devices sold in Malaysia must carry local labelling as per requirement from the Medical Device Authority. To allow this to happen, the packaging of the non-refrigerated devices will be opened and resealed for labelling purposes.
All medical devices sold in Malaysia must carry local labelling as per requirement from the Medical Device Authority. To allow this to happen, the packaging of the non-refrigerated devices will be opened and resealed for labelling purposes.
Product Information
- rotary knobs, non-refrigerated, without rotor
- 230 V/50 – 60 Hz (GB)

You will find additional download material at the bottom of this page
Features
- with rotary knob control panel for quick parameter changes
- without rotor
- non-refrigerated
- 230 V, 50 – 60 Hz