MENU
BR | BRL
BR | BRL
-
- Todas as centrífugas
- Centrífugas de bancada
- Centrífugas de chão
- Centrífugas refrigeradas
- Microcentrífugas
- Centrífugas multifuncionais
- Centrífugas de alta velocidade
- Ultracentrífugas
- Concentrador
- Consumíveis de alta velocidade e de ultracentrifugação
- Tubos de centrifugação
- Placas de centrifugação
- Software de gestão de equipamentos
- Gestão de amostras e informações
-
- Todas as pipetas, dispensadores e equipamentos para manuseio de líquidos automatizados
- Pipetas mecânicas
- Pipetas eletrônicas
- Pipetas multicanal
- Pipetas de deslocamento positivo e dispensadores
- Ponteiras de pipeta
- Dispensadores de frascos
- Controladores de pipeta
- Acessórios para dispensadores e pipetas
- Pipetagem Automatizada
- Consumíveis de automação
- Acessórios de automação
- Serviços para pipetas e equipamentos para manuseio de líquidos
Sorry, we couldn't find anything on our website containing your search term.
Sorry, we couldn't find anything on our website containing your search term.

Vaccine Research Discovered
Well thought-out tools for pharmaceutical and biotech laboratories
Read more
Read less
STRONG GROWING VACCINES
Protein subunit vaccines
Protein subunit vaccines (e.g., for influenza, hepatitis B and C) are a proven strategy for decades and still today in the spotlight for pandemic management.
➕ Require no production of the whole pathogen because the vaccine consists of immunogenic viral or bacterial fragments generated by recombinant protein techniques. Since non-pathogenic, they are considered as safe. Multiple expression systems for antigenic protein components such as yeast, bacteria, insect, mammalian, and plant cells, allow the production of large quantities and to address unmet needs.
➖ Establishment of a proper scale-up of antigen production can be a major constraint. Several booster doses and adjuvants are needed.
Protein subunit vaccines (e.g., for influenza, hepatitis B and C) are a proven strategy for decades and still today in the spotlight for pandemic management.
➕ Require no production of the whole pathogen because the vaccine consists of immunogenic viral or bacterial fragments generated by recombinant protein techniques. Since non-pathogenic, they are considered as safe. Multiple expression systems for antigenic protein components such as yeast, bacteria, insect, mammalian, and plant cells, allow the production of large quantities and to address unmet needs.
➖ Establishment of a proper scale-up of antigen production can be a major constraint. Several booster doses and adjuvants are needed.
Ler mais
Virus-like particle vaccines
Virus-like particle vaccines are self-assembled viral structural proteins that mimic the native virus. This feature enables to trigger strong immune responses.
➕ Lacking the viral genome, the safety profile is excellent. The production is scalable and safe as no living virus or inactivation steps are involved.
➖ The particle assembly is sometimes challenging, the manufacturing process complicated.
Virus-like particle vaccines are self-assembled viral structural proteins that mimic the native virus. This feature enables to trigger strong immune responses.
➕ Lacking the viral genome, the safety profile is excellent. The production is scalable and safe as no living virus or inactivation steps are involved.
➖ The particle assembly is sometimes challenging, the manufacturing process complicated.
Ler mais
IMPROVE YOUR RECOMBINANT PROTEIN VACCINE DISCOVERY
Ler mais
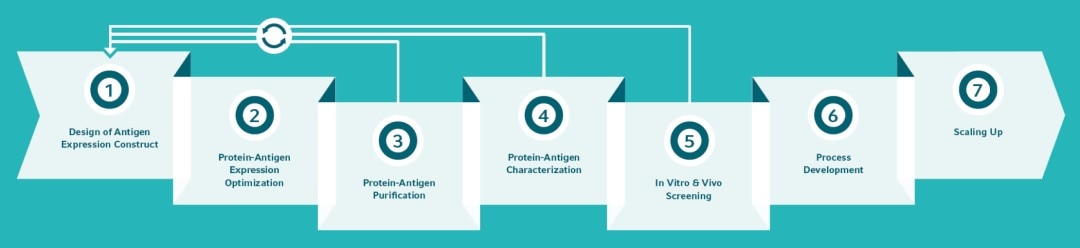
*All products are for Research Use Only (RUO).
Ler mais
DOWNLOADS

Application Note

Application Note

White Paper
Ler mais