MENU
IT | EUR
-
-
-
-
- WHX Labs Dubai 2026
- analytica 2026
- Forum Labo 2026
- SLAS Europe 2026
- Forum Labo 2026 Bioprocess
- Bioprocessing Summit Europe 2026
- BioProcess International 2026
- Swiss Biotech Day 2025
- ISCT 2026
- Pichia 2026
- Future Labs Live 2026
- BioProScale Sympposium 2026
- ASGCT 2026
- Eppendorf Automation Forum 2026
- ESACT 2026
-
-
-
-
- WHX Labs Dubai 2026
- analytica 2026
- Forum Labo 2026
- SLAS Europe 2026
- Forum Labo 2026 Bioprocess
- Bioprocessing Summit Europe 2026
- BioProcess International 2026
- Swiss Biotech Day 2025
- ISCT 2026
- Pichia 2026
- Future Labs Live 2026
- BioProScale Sympposium 2026
- ASGCT 2026
- Eppendorf Automation Forum 2026
- ESACT 2026
IT | EUR
-
- Tutte le centrifughe
- Centrifughe da banco
- Centrifughe a pavimento
- Centrifughe refrigerate
- Microcentrifughe
- Centrifuga polivalente
- Centrifughe ad alta velocità
- Ultracentrifughe
- Concentratore
- Prodotti IVD
- Consumabili per centrifughe ad alta velocità e ultracentrifughe
- Provette per centrifughe
- Piastre per centrifughe
- Software di gestione degli apparecchi
- Gestione dei campioni e delle informazioni
-
- Tutte le pipette, i dispenser e i sistemi automatizzati per la manipolazione dei liquidi
- Pipette meccaniche
- Pipette elettroniche
- Pipette multicanale
- Pipette a spostamento positivo e dispenser
- Puntali per pipette
- Dispenser per flaconi
- Ausili di pipettaggio
- Accessori per dispenser e pipette
- Pipettaggio automatizzato
- Consumabili per l'automazione
- Accessori per l'automazione
- Servizi per sistemi di manipolazione dei liquidi e pipette
Sorry, we couldn't find anything on our website containing your search term.
Sorry, we couldn't find anything on our website containing your search term.

Vaccine Research Discovered
Well thought-out tools for pharmaceutical and biotech laboratories
Read more
Read less
STRONG GROWING VACCINES
Protein subunit vaccines
Protein subunit vaccines (e.g., for influenza, hepatitis B and C) are a proven strategy for decades and still today in the spotlight for pandemic management.
➕ Require no production of the whole pathogen because the vaccine consists of immunogenic viral or bacterial fragments generated by recombinant protein techniques. Since non-pathogenic, they are considered as safe. Multiple expression systems for antigenic protein components such as yeast, bacteria, insect, mammalian, and plant cells, allow the production of large quantities and to address unmet needs.
➖ Establishment of a proper scale-up of antigen production can be a major constraint. Several booster doses and adjuvants are needed.
Protein subunit vaccines (e.g., for influenza, hepatitis B and C) are a proven strategy for decades and still today in the spotlight for pandemic management.
➕ Require no production of the whole pathogen because the vaccine consists of immunogenic viral or bacterial fragments generated by recombinant protein techniques. Since non-pathogenic, they are considered as safe. Multiple expression systems for antigenic protein components such as yeast, bacteria, insect, mammalian, and plant cells, allow the production of large quantities and to address unmet needs.
➖ Establishment of a proper scale-up of antigen production can be a major constraint. Several booster doses and adjuvants are needed.
Approfondisci
Virus-like particle vaccines
Virus-like particle vaccines are self-assembled viral structural proteins that mimic the native virus. This feature enables to trigger strong immune responses.
➕ Lacking the viral genome, the safety profile is excellent. The production is scalable and safe as no living virus or inactivation steps are involved.
➖ The particle assembly is sometimes challenging, the manufacturing process complicated.
Virus-like particle vaccines are self-assembled viral structural proteins that mimic the native virus. This feature enables to trigger strong immune responses.
➕ Lacking the viral genome, the safety profile is excellent. The production is scalable and safe as no living virus or inactivation steps are involved.
➖ The particle assembly is sometimes challenging, the manufacturing process complicated.
Approfondisci
IMPROVE YOUR RECOMBINANT PROTEIN VACCINE DISCOVERY
Approfondisci
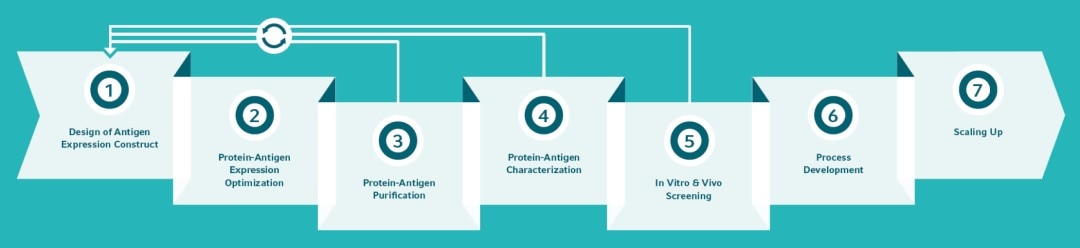
*All products are for Research Use Only (RUO).
Approfondisci
DOWNLOADS

Application Note

Application Note

White Paper
Approfondisci