メニュー
JP | JPY
-
-
-
- Challenges and Chances: A Review of the 1st Stem Cell Community Day
- Summertime, and the Livin’ Is Easy…
- Follow-on-Biologics – More than Simple Generics
- Bacteria Versus Body Cells: A 1:1 Tie
- Behind the Crime Scene: How Biological Traces Can Help to Convict Offenders
- Every 3 Seconds Someone in the World Is Affected by Alzheimer's
- HIV – It’s Still Not Under Control…
- How Many Will Be Convicted This Time?
- Malaria – the Battle is Not Lost
- Physicians on Standby: The Annual Flu Season Can Be Serious
- At the Forefront in Fighting Cancer
- Molecular Motors: Think Small and yet Smaller Again…
- Liquid Biopsy: Novel Methods May Ease Cancer Detection and Therapy
- They Are Invisible, Sneaky and Disgusting – But Today It’s Their Special Day!
- How Many Cells Are in Your Body? Probably More Than You Think!
- What You Need to Know about Antibiotic Resistance – Findings, Facts and Good Intentions
- Why Do Old Men Have Big Ears?
- The Condemned Live Longer: A Potential Paradigm Shift in Genetics
- From Research to Commerce
- Chronobiology – How the Cold Seasons Influence Our Biorhythms
- Taskforce Microbots: Targeted Treatment from Inside the Body
- Eyes on Cancer Therapy
-
-
-
-
-
- Challenges and Chances: A Review of the 1st Stem Cell Community Day
- Summertime, and the Livin’ Is Easy…
- Follow-on-Biologics – More than Simple Generics
- Bacteria Versus Body Cells: A 1:1 Tie
- Behind the Crime Scene: How Biological Traces Can Help to Convict Offenders
- Every 3 Seconds Someone in the World Is Affected by Alzheimer's
- HIV – It’s Still Not Under Control…
- How Many Will Be Convicted This Time?
- Malaria – the Battle is Not Lost
- Physicians on Standby: The Annual Flu Season Can Be Serious
- At the Forefront in Fighting Cancer
- Molecular Motors: Think Small and yet Smaller Again…
- Liquid Biopsy: Novel Methods May Ease Cancer Detection and Therapy
- They Are Invisible, Sneaky and Disgusting – But Today It’s Their Special Day!
- How Many Cells Are in Your Body? Probably More Than You Think!
- What You Need to Know about Antibiotic Resistance – Findings, Facts and Good Intentions
- Why Do Old Men Have Big Ears?
- The Condemned Live Longer: A Potential Paradigm Shift in Genetics
- From Research to Commerce
- Chronobiology – How the Cold Seasons Influence Our Biorhythms
- Taskforce Microbots: Targeted Treatment from Inside the Body
- Eyes on Cancer Therapy
-
-
JP | JPY
Sorry, we couldn't find anything on our website containing your search term.
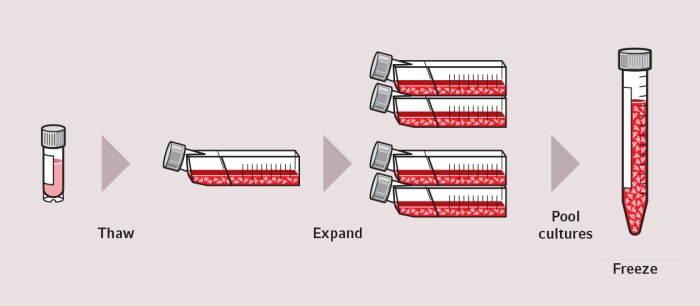
How to thaw cells – Guide for more reproducible cryopreservation results
Lab Academy
- 細胞生物学
- 細胞培養
- 実験室の日常業務
- 汚染
- 幹細胞
- 細胞培養用消耗品
- CO₂インキュベーター
- エッセー
Cell thawing is an essential and sometimes undervalued element in all areas of cell-based research and production. A standardized cryopreservation process – including a standardized cell thawing protocol – helps to use cell stocks optimally for reproducible, reliable results.
Our White Paper “ ” highlights different aspects of the cell thawing process and includes the following chapters:
- Introduction
- How to thaw cells - Basics
- Cell thawing methods – comparison of different methods
- Considerations prior to selecting a cell thawing method
- Summary
もっと読む
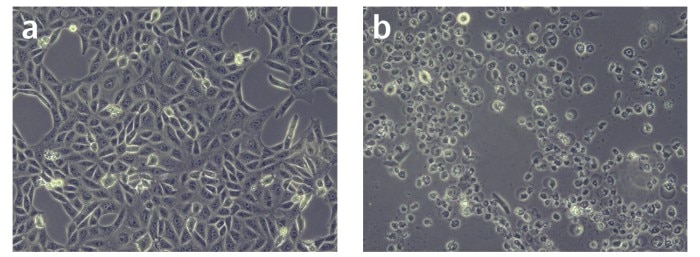
Incorrect cell thawing can lead to abnormalities in cell shape after initial seeding. Example of normal (a) and abnormal (b) morphology of Vero cells (10 x).
How to thaw cells - Basics
Cell freezing and thawing require different rates of temperature change to ensure high cell viability. Cell freezing needs to occur at a slow, controlled cooling rate. In contrast, cell thawing works best when it is done quickly because the disappearance of ice around the cell does not have the same damaging effects as ice formation during cryopreservation. Therefore, it is preferable to bring the cells back to normal culture conditions as soon as possible, where (in the case of adherent cells) they can anchor to a surface.
Usually, a typical cell thawing protocol begins with removing cryovials from the liquid nitrogen tank. Here, it is important to be familiar with standard precautions for working with liquid nitrogen. Additionally, if a cryovial is not sealed properly and was stored in the liquid phase, liquid nitrogen may have seeped into the cryovial over time. This leads to a rapid pressure build-up inside the vial shortly after removing it from the liquid nitrogen tank. Therefore, wearing adequate face protection is necessary.
Usually, a typical cell thawing protocol begins with removing cryovials from the liquid nitrogen tank. Here, it is important to be familiar with standard precautions for working with liquid nitrogen. Additionally, if a cryovial is not sealed properly and was stored in the liquid phase, liquid nitrogen may have seeped into the cryovial over time. This leads to a rapid pressure build-up inside the vial shortly after removing it from the liquid nitrogen tank. Therefore, wearing adequate face protection is necessary.
もっと読む

Image source: Minerva Studio/shutterstock.com
Next, the cell suspension inside the vial must be thawed. A frequently used rule of thumb is that upon beginning the cell thawing process in a standard cryovial with 1 mL cell suspension, all ice needs to have disappeared within a few minutes. Rapid heating of the cell suspension prevents localized recrystallization during cell thawing, which can cause cellular damage [1]. Sometimes thawing cells within a few minutes is not possible, e.g. due to time spent to find the exact vial or due to distance between the location of the liquid nitrogen storage and the thawing location. In this case, it is preferable to leave the cryovials at the lowest possible temperature followed by rapid thawing instead of both slow thawing and leaving the cells in thawed freezing medium for longer than necessary [2].
When all ice has disappeared, any further adverse effects of the cryoprotective agent on the cells need to be minimized by two possible ways. Cells can be either cultured directly after thawing, for example in a T-flask, while ensuring the freezing medium is diluted by at least a factor of ten with normal cell culture medium. Alternatively, it is possible to dilute the freezing medium with normal cell culture medium, centrifuge the tube, discard the medium, and resuspend the thawed cells in fresh culture medium [2, 3, 4]. To check that the cell thawing protocol was successful, it is recommended to determine the percentage of viable cells (e.g. with a trypan blue staining and a cell counter) [5].
To improve cell thawing standardization, it is good practice to monitor the cell culture over several days for any abnormalities in shape or growth rate (see image at the top for an example). Poor or inconsistent cell growth can be a sign of cell stock problems or problems within the cryopreservation process. Therefore, early detection can help to minimize experimental errors.
Different methods and equipment exist to thaw cells in cryovials – water bath, hand-warming, bead bath, and dedicated devices.
Read more about the advantages and disadvantages of different cell thawing methods, as well as lab- and cell-type specific considerations in our White Paper “Cell Thawing Protocol Standardization - Guide for More Reproducible Cryopreservation Results” -
When all ice has disappeared, any further adverse effects of the cryoprotective agent on the cells need to be minimized by two possible ways. Cells can be either cultured directly after thawing, for example in a T-flask, while ensuring the freezing medium is diluted by at least a factor of ten with normal cell culture medium. Alternatively, it is possible to dilute the freezing medium with normal cell culture medium, centrifuge the tube, discard the medium, and resuspend the thawed cells in fresh culture medium [2, 3, 4]. To check that the cell thawing protocol was successful, it is recommended to determine the percentage of viable cells (e.g. with a trypan blue staining and a cell counter) [5].
To improve cell thawing standardization, it is good practice to monitor the cell culture over several days for any abnormalities in shape or growth rate (see image at the top for an example). Poor or inconsistent cell growth can be a sign of cell stock problems or problems within the cryopreservation process. Therefore, early detection can help to minimize experimental errors.
How to thaw cells – Advantages and disadvantages of different methods
Different methods and equipment exist to thaw cells in cryovials – water bath, hand-warming, bead bath, and dedicated devices. Read more about the advantages and disadvantages of different cell thawing methods, as well as lab- and cell-type specific considerations in our White Paper “Cell Thawing Protocol Standardization - Guide for More Reproducible Cryopreservation Results” -
もっと読む
References
[1] Hunt, CJ. Technical Considerations in the Freezing, Low-Temperature Storage and Thawing of Stem Cells for Cellular Therapies. Transfusion Medicine and Hemotherapy 2019; 46: 134–149.
[2] Baust, JM et al. Best practices for cryopreserving, thawing, recovering, and assessing cells. In Vitro Cellular & Developmental Biology – Animal 2017.
[3] www.lgcstandards-atcc.org/Documents/Marketing_Literature/Animal_Cell_Culture_Guide/Cryopreservation.aspx
[4] www.lgcstandards-atcc.org/How_to_Revive_Cultures.aspx
[5] Strober, W. Trypan Blue Exclusion Test of Cell Viability. Current Protocols in Immunology 2019.
[2] Baust, JM et al. Best practices for cryopreserving, thawing, recovering, and assessing cells. In Vitro Cellular & Developmental Biology – Animal 2017.
[3] www.lgcstandards-atcc.org/Documents/Marketing_Literature/Animal_Cell_Culture_Guide/Cryopreservation.aspx
[4] www.lgcstandards-atcc.org/How_to_Revive_Cultures.aspx
[5] Strober, W. Trypan Blue Exclusion Test of Cell Viability. Current Protocols in Immunology 2019.
もっと読む
Related documents

White Paper No.60
PDF
